Reasonable Expectation of Success in Treating Different Conditions Not Implied by Structural Similarity
- February 7, 2023
- Article
Associated People
Associated Technologies
In the matter of Ex parte Taleb, et al., the Patent Trial and Appeal Board (PTAB) determined on January 31, 2023, that an examiner (James Anderson, supported by Supervisory Patent Examiners, Jeffrey Lundgren and Kortney Klinkel) failed to establish the obviousness of claims by failing to establish a reasonable expectation of success in achieving a claimed result of a method claim. The claim in question recited
[a] method of treating obesity in an obese subject in need thereof, comprising administering to the obese subject a therapeutically effective amount of an indoleamine 2,3-dioxygenase (IDO) inhibitor selected from the group consisting of β-(3-benzofuranyl)-alanine, β-(3-benzo(b)thienyl)-alanine, 6-nitro-tryptophan, 6-fluoro-tryptophan, 4-methyl-tryptophan, 5-methyl tryptophan, 6-methyl-tryptophan, 5-methoxytryptophan, 5-hydroxy-tryptophan, 3,3'-diindolylmethane, 5-Br-4-Cl-indoxyl 1,3-diacetate, 9-vinylcarbazole, acemetacin, 5-bromo-tryptophan, 5-bromoindoxyl diacetate, pyrrolidone dithiocarbamate, and 4-phenylimidazole.
The structures of the claimed compounds are provided below:
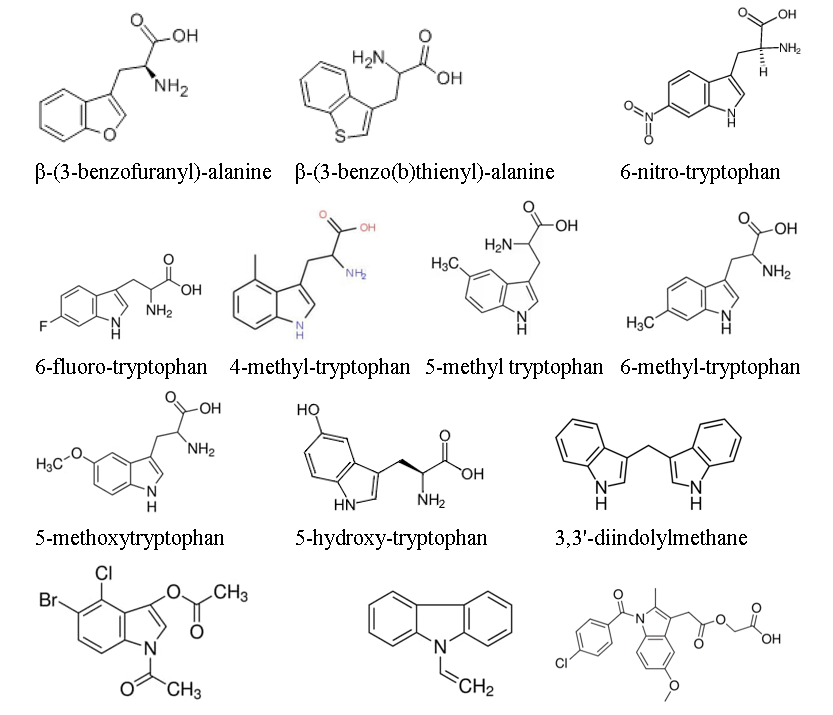
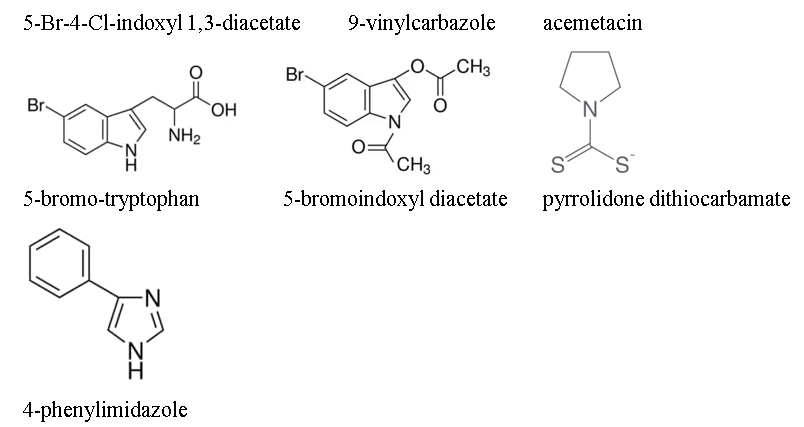
The original filing of the appealed application presented four claims, three of which were independent. Original claim 1 recited “[a] method of treating obesity in a subject in need thereof comprising administering to the subject a therapeutically effective amount of an indoleamine[-2,3-]dioxygenase (IDO) inhibitor,” while independent claims 2 and 4 respectively recited methods of “for the prophylactic treatment of an obesity related disease” and “improving insulin sensitivity in an obese subject.”
After novelty rejections of the claims, the applicant amended the claims to recite specific indoleamine-2,3-dioxygenase (IDO) inhibitors, which were further defined after three further office actions containing obviousness rejections, relying at the outset on the primary and tertiary prior art references appealed. The tertiary prior art reference related to the efficacy of 1-methyl tryptophan in reversing IDO-mediated arrest of T-cell proliferation in human epithelial ovarian cancer. The narrowing of the IDO inhibitors eliminated, among other things, 1-methyl tryptophan from the claims.
The rejection of the applicant’s appealed method claim relied upon a combination of five (5) prior art references. The examiner argued that the primary non-patent literature (NPL) reference described several salient details related to treating obesity, namely:
(1) morbid obesity is associated with low-grade systemic inflammation and immune activation, which results in the synthesis and release of pro-inflammatory cytokines, including TNF-α and IFN-γ;
(2) elevated production of TFN-α induces expression of indoleamine 2,3-dioxygenase (IDO), which functions to convert tryptophan into kynurenine;
(3) lowered tryptophan availability from increased kynurenine production affects serotonin production, since tryptophan is a precursor in the synthesis of serotonin, thereby resulting in diminished serotonin in the central nervous system;
(4) decreased serotonin levels increase the susceptibility to mood disturbance, depression and increased appetite; and
(5) quantitative serotonin deficiency can lead to a reward deficiency syndrome that perpetuates pathologic eating, thereby supporting a functional link to obesity.
The examiner relied on a secondary NPL reference for its disclosure of a study of obese and non-obese adolescents finding that serum TNF-α was significantly higher in the obese group than the non-obese group. A tertiary NPL reference relied upon by the examiner described that levo-1-methyl-tryptophan, i.e.,
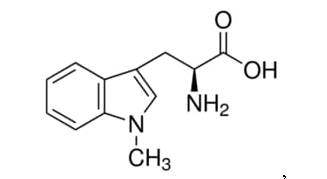
is an IDO inhibitor and a quaternary NPL reference gave the definition of overweight as a BMI of 25.0 to 29.9. The fifth prior art NPL reference was relied on for a disclosure that 1-methyl-tryptophan and (among others) several other recited compounds, i.e., β-(3-benzofuranyl)-DL-alanine, β-(3-benzo(b)thienyl)-DL-alanine, 6-nitro-L-tryptophan, 3,3'-diindolylmethane, 5-Br-4-Cl-indoxyl 1,3-diacetate, 9-vinylcarbazole, acemetacin, 5-bromo-DL-tryptophan, and 5-bromoindoxyl diacetate, are IDO inhibitors.
The applicant had initially also claimed 1-methyl-tryptophan, but removed 1-methyl-tryptophan from the claim to attempt to define over the combined cited art. The applicant argued the lack of predictability of maintaining the effect of 1-methyl-tryptophan using the different active ingredients from the fifth prior art NPL reference, which:
cites various IDO inhibitors but does not teach intervention in inflammatory disease or obesity and/or obesity-related diseases. Thus, the biological outcome of the use of each as a treatment for obesity and/or obesity-related disorders cannot be predicted based on the chemical structures of the various inhibitors. One of ordinary skill in the art would recognize that minor changes in constituents of a family chemical structures can dramatically change the binding and efficacy between those family members. Therefore, it is not obvious that IDO inhibitors effective for neurological disorders would also be effective for treating obesity and/or obesity-related disorders.
The examiner maintained on appeal that it would have been obvious to administer an IDO inhibitor to treat or inhibit obesity because (i) the primary NPL reference teaches that immune activation resulting in increased TNF-α and systemic inflammation in obesity causes elevated expression of IDO, an effect that reduces circulating serotonin by utilizing tryptophan - a serotonin synthesis precursor - for forming kynurenine, thereby increasing appetite, and (ii) the secondary NPL reference teaches that elevated TNF-α is observed in overweight subjects. The examiner felt that this established that overweight subjects would exhibit elevated IDO expression as a result of increased TNF-α.
The examiner argued that inhibiting IDO would have been expected to minimize tryptophan depletion, thereby promoting serotonin synthesis, which increase serotonin. Deficiency in serotonin was known to lead to pathologic eating due to increased appetite, thus the examiner argued that increasing serotonin levels would have reasonably expected to inhibit obesity in such subjects.
The applicant / appellant argued that the primary NPL reference teaches that elevated production of IFN-γ induces expression of IDO, and that TNF-α is expressed because of IDO expression, not as a cause, and further that the primary NPL reference teaches that leptin, ghrelin, TGF-β, IL-1, IL-6, IL-8, IL-10, haptoglobin, factors of the complement cascade, chemoattractant cytokines, neopterin, cyclohydrolase, all play a role in the chronic immune activation that affects obesity. That is, the applicant / appellant argued the opposite effect would have been expected by IDO inhibition, and that many factors besides IDO affected obesity.
The PTAB found that the primary prior art NPL reference in fact stated that IDO-mediated tryptophan degradation due to chronic immune activation can be considered as the driving force for food intake in obese subjects, but that this suggestion depended on a string of inferences. The PTAB noted the examiner’s citations, but pointed to the conclusion in the primary NPL reference that
[a]ccumulating evidence suggests that IDO might be an important contributing factor to the complex pathophysiology [sic] of morbid obesity that links the immune system with central regulators of hunger, satiety and mood disturbances and thus can be considered as a novel scientific target that deserves further investigation in the urgent need to treat overweight [subjects].
The PTAB found that the primary prior art NPL reference described a hypothesis connecting IDO activity to obesity, but lacked evidence supporting the theory that increased IDO activity causesobesity, and provides no evidence showing that IDO expression or activity is higher in obese subjects than in lean subjects, or that increased IDO activity causes decreased serotonin levels in obese subjects, or that lower serotonin levels cause subjects to overeat, or that inhibiting IDO activity causes serotonin levels to rise and, ultimately, causes subject to eat less and lose weight.
The PTAB found that no other cited reference established this link, and that the fifth prior art NPL reference merely showed the treatment of other conditions by IDO inhibition. Therefore the PTAB was persuaded that there was no reasonable expectation of successfully treating obesity or obesity-related symptoms with the compounds in the appealed claims. Accordingly, the obviousness rejection was reversed.
This case is a reminder that, particularly for method claims in which a specific medicinal or therapeutic effect is claimed, structural similarity is generally insufficient to extrapolate efficacy to structurally related compounds for which that medicinal or therapeutic effect is unknown. The pharmaceutical chemical art is still treated as unpredictable by the PTAB, even if the examining corps does not as deferentially consider this fact.










 Counseling & Strategic Advice
Counseling & Strategic Advice IP Transactions
IP Transactions Litigation
Litigation PTAB Proceedings
PTAB Proceedings Technology Transfer
Technology Transfer Trademark & Designs
Trademark & Designs U.S. Patent Procurement (Application Drafting & Prosecution)
U.S. Patent Procurement (Application Drafting & Prosecution)








